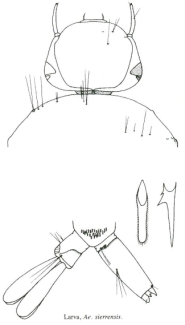
Aëdes is the Greek word for disagreeable. Without the dieresis the word means house or building. Although Meigen did not use a dieresis, he translated it as troublesome. Some authorities, therefore, write the generic name Aëdes. Most species of British Columbian mosquitoes belong to this genus. The females all have short palps, usually less than one quarter of the length of the proboscis, and in both sexes the posterior margin of the scutellum is tri-lobed with the setae in three tufts.
Aedes is a large and variable genus and in the field the most reliable character to separate females from other mosquito genera is the pointed abdomen. Males can be identified in the field by their large and separated gonocoxites but if these are not obvious the thorax can be examined for the presence of postspiracular setae which are absent in the males of Culex, Culiseta, and Mansonia. A slide of the terminalia, as well as confirming the genus, can be used to determine the species. (See Wood et at. 1979).
When at the water surface, the larvae of all culicines hang downwards from the hydrophobic tip of the siphon and are thus easily distinguished from anophelines.
Aedes larvae can be distinguished from those of Culex and Culiseta by the position of the siphon seta (1-S). It is never at the base of the siphon in aedines and can be seen with a hand lens if the larva cooperates.
The pupae are hard to identify. It is usually simpler to let them emerge.
Nearly all aedine adults in British Columbia die in late summer or autumn. The eggs are laid singly or in clusters, usually in crevices at the margins of suitable breeding sites. They do not float. Most aedines overwinter as eggs.
|
|