Being the second largest deer in British Columbia, the Elk is much larger than the Woodland Caribou, White-tailed Deer and Mule Deer. The Moose is the closest in size, but it has a dark body with lighter coloured legs, and a long head and bulbous nose. In contrast, the Elk has a fawn or reddish-brown body with darker legs and neck, a compact, dark hairless nose, and a large light-coloured rump patch. The Elk’s antlers also differ markedly from the palmated antlers of the Moose. At maturity, Elk antlers typically have a long main beam growing up and back from the head, with up to six long, pointed tines on each beam.
The next most similar species to Elk is Woodland Caribou, but again, coloration and antlers differ. The Elk is reddish brown not dark greyish or chocolate brown as is the Caribou, and has a dark neck compared to the light neck of the Caribou. The Elk also lacks the white ring of hair above the hooves and does not make a clicking sound when walking. Finally, female Caribou often have small antlers while female Elk almost never grow them, and antlers of adult male Caribou have palmated tines with rounded ends unlike the sharp pointed ones of Elk.
In skulls, the presence of upper canine teeth indicates either an Elk or a Caribou. But Elk canines are much larger, have rounded ends and project further below the premaxillae (and gum) than those of Caribou. Also unlike Caribou, the premaxilla in the Elk skull contacts the nasal bone, and when viewed from above, the shape is rounded, lacking the angular profile of Caribou. Finally, in an Elk skull the vomer does not divide the posterior nares as it does in the Caribou skull.
Elk tracks are slightly smaller but less pointed at the front than those of Moose, and are proportionally narrower than those of Caribou, Cattle or Bison. Except for the soft spring faeces, dry adult Elk pellets are usually dimpled at one end and have a small projection at the other, giving them an almost acorn shape. Pellets of Odocoilid deer are about half the size and lack these end characteristics. Moose pellets are about the same size as those of Elk but they are usually rounded at either end.
|
Gestation lasts about 255 days, and just before giving birth, females often leave the herd to have their young, sometimes staying secluded for up to two weeks. Females usually bear one young weighing 10 to 15 kg, in late May or early June. Twins are rare. For the first few weeks of life, the young are hiders (see page 46 in General Biology), usually resting along the forest edge or in other vegetation cover, while the remainder of the herd forages nearby. When in danger, a young Elk makes a high screaming sound to bring its mother running to its rescue.
Most females give birth to their first young on their third birthday, but in some populations it may be a year earlier. Males can produce sperm at 1.5 to 2 years, but usually do not become fully sexually active and take part in the rut until at least 3 years old, and for most, not until they are about 7 or 8.
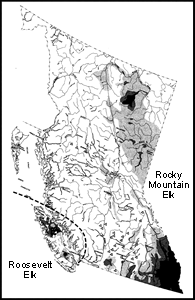
| Elk are generalist herbivores with a mixed browser-grazer diet and a high degree of dietary flexibility. Consequently, Elk tend to show diet overlap with many other species of ungulates. The relative importance of forages varies with subspecies, season and even the herd. Rocky Mountain Elk consume a variety of grasses, sedges and forbs, as well as shrubs such as willows, Soopolallie, Saskatoon and Trembling Aspen. Their winter diets are primarily grasses, sedges and shrubs, with forbs becoming important in spring and summer. Rocky Mountain Elk probably eat more grasses, sedges and forbs than Roosevelt Elk do, and use more open grass-shrub communities. Roosevelt Elk, on the other hand, live mainly in more heavily forested habitats, which is reflected in their diet. Important browse species for Roosevelt Elk include Red Elderberry, various Vaccinium and Rubusspecies, willows, Dull Oregon-grape, Pacific Ninebark, Amabalis Fir and Western Hemlock. These Elk can also have major impacts on newly planted tree seedlings such as Western Red-cedar. Roosevelt Elk also eat various grasses and sedges, and many types of forbs, such as Skunk Cabbage, in spring and summer. Sword Ferns and Deer Ferns are also important forages in most seasons except summer. Preferred forages used by both subspecies grow in early successional stages following a fire, or in young cut-overs following logging. Riparian areas are also important for foraging, especially for Roosevelt Elk. When not hunted, Elk adapt well to humans and find lawns and golf courses excellent places to graze. |
An Elk’s age can be determined by tooth succession, and by cementum annuli counts using the first incisor or first lower molar; tooth wear is not always a reliable method. Elk can be long-lived, but the average longevity varies between populations. Females usually live longer than males, reaching 20 to 24 years compared to 12 to 14 years for males.
|
Wolves are the main predators of all age-sex classes of Elk. Cougars also prey on Elk, and both Black and Grizzly bears prey on young of the year. A nematode worm, Dictyocaulus sp., may be a significant parasite for Rocky Mountain Elk, especially during periods of poor nutrition. Domestic Cattle is the primary host for this parasite, although it is also reported in Odocoilid deer. The parasite lives in the air passages, and when present in large numbers, can make breathing difficult. This can lead to respiratory infection, with or without other disease organisms, and result in bronchitis. The Winter Tick can be a serious parasite of Elk, especially when severe winters and shortage of winter range contributes to malnutrition. The large American Liver Fluke is common in populations of both subspecies of Elk in B.C.; it seems to have little effect on Elk, but it can kill Moose and domestic ungulates. The life cycle of the Liver Fluke involves aquatic snails, so the fluke is rarely seen in regions with predominantly dry habitats. The liver of any infected animal may have large cavities and tracks filled with immature flukes, surrounded by quantities of soft, blackish material. Throughout the range of Elk, severe winters with high snowfall can lead to malnutrition and increased mortality either from starvation or increased susceptibility to predation.
|
Elk are highly social animals that congregate in large maternal groups. But, as with most hoofed mammals in B.C., the mature males and females live apart for most of the year. Males leave their maternal groups when 2.5 to 3 years old and socialize with females only during the mating season.
Maternal groups can be quite large, and their size depends in part on habitat; larger groups tend to develop in more open areas. Adult female and juvenile Rocky Mountain Elk form the largest groups, but on Vancouver Island, maternal groups of Roosevelt Elk can number more than 100. Such large groups are not necessarily stable and may separate into smaller units. All-male groups of similarly aged individuals are much smaller and may be even less stable over time. While feeding or walking in groups, Elk often make a low grunting call, perhaps to maintain contact among members.
The mating or rutting period usually begins in September and extends into October. Males are especially active, and perhaps their best known and most obvious behaviour is their bugling vocalization. This hollow squealing whistle starts with a two-note call, extends into a long higher note that drops suddenly in pitch, and usually ends with two or more separate short whistles or grunts. While bugling, the male stands with his mouth open, and the last short calls are usually accompanied by sharp contractions of the belly, and often by spurts of urine. These calls appear to serve as challenges to other males and warnings to stay away. Male Elk also threaten each other by approaching an opponent with head and nose raised and the upper lip curled to reveal the small canine teeth, while making a hissing sound. Besides the bugling and head-up threat, an adult male will make noises and threats by thrashing bushes and rubbing small trees with his antlers, leaving vertical gouges edged by frayed bark. A male Elk will also paw the ground and urinate on it, then wallow on the wet ground. Soaking his mane and body with urine increases his body odour, which probably has signal value to other males as well as to females. (Wet wallows made during the rut are different from the drier ones used in summer, which Elk use to discourage biting insects. Elk may rub their antlers against small trees and saplings at other times of the year as well, and signs of this activity can be concentrated in some areas.)
Even before the rut begins in earnest, males will spar and fight with each other. Large males of similar size often trot or walk beside each other a few metres apart for up to 200 metres, before turning and repeating the parallel walk. During these walks, they may tip their antlers towards each other, which sometimes leads to a fight. They fight head to head after engaging their antlers, pushing and twisting as they try to throw each other off balance so they can try to gore their opponent with their antler points. When fighting head to head, the long brow tine helps protect the face and eyes from damage, but antler points can break. Serious fights between equally matched males can sometimes lead to injury and, occasionally, to the death of one. Females – and males without hard antlers – fight by rearing up on their hind legs, ears back, flaying their front legs rapidly as they strike at their opponent with the front hooves. Sometimes just holding the head high with ears back is a sufficient threat to end a dispute.
Elk are polygamous, and adult males defend a harem. Most harems consist of one adult male and up to 20 (or more) adult females and young. The harem male excludes all other males except calves. He must constantly defend the females from the satellite males that hang around the outer edges of the harem trying to steal females or quickly copulate with them. He chases away rivals by bugling and using the threats described above. The harem male has little control over the movements of the females. He can only try to keep the group together by using a specific herding posture. He circles a female from her rear or side in a broadside display, head low but with the nose tipped up, so the antlers are alongside his neck. If the female does not respond and move back into the group, the male usually rushes at her bringing his nose down rapidly so the antlers swing forward and down to push or bump her rump.
Courtship patterns are not particularly elaborate in Elk. The male’s courtship approach is the opposite of the herding posture. The courting male Elk moves towards the female from the front with his head and antlers raised slightly, flickering his tongue and making licking or soft grunting sounds. The female often responds by lowering her head, neck extended, sometimes ears back, while making biting movements opening and closing her mouth rapidly. The courting male sniffs her rear, lip-curls, and puts his neck over her rump. Like most deer, copulation takes place during a brief copulatory jump, in which the female stands still and the male grasps her with his forelegs just in front of her pelvis. He makes a single thrusting ejaculatory jump with the head thrown back and the hind feet often leaving the ground momentarily.
|
|