Two subspecies of Caribou have been recognized in British Columbia.
Subspecies: Woodland Caribou (Rangifer tarandus caribou)
Three populations of this subspecies are found in BC (there are 52 across Canada), representing three ecotypes (Mountain Caribou, Boreal Woodland Caribou, and Northern Woodland Caribou). All are of conservation concern.
Subspecies Description
Woodland Caribou, a medium to large deer, is the largest of all Caribou. Males can weigh as much as 300 kg and females up to 130 kg, but there are few weights available for this deer in the province. Woodland Caribou is also the darkest subspecies of any in the world. The body is usually a dark, almost chocolate brown, as is the face, belly and legs.
Woodland Caribou has the heaviest antlers of all Caribou, although they are not the longest, and their spread is often narrower. Though Woodland Caribou have the general antler plan of the species, their antlers do vary: they can be short, heavy and strongly palmated, or long with only a slightly palmated terminal section.
Measurements
total length: male: 2,327 mm (2,060-2,980) n = 15; female: 2,007 mm (1,760-2,200) n = 70
tail vertebrae: male: 177 mm (130-220) n = 10; female: 142 mm (50-220) n = 51
hind foot: male: 548 mm (430-660) n = 10; female: 528 mm (380-660) n = 48
ear: male: 149 mm (130-160) n = 6; female: 143 mm (120-160) n = 15
shoulder: male: 1,384 mm (1,245-1,580) n = 14; female: 1,222 mm (1,030-1,390) n = 43
chest male: 1,447 mm (1,390-1,530) n = 12; female:1,277 mm (1,180-1,440) n = 75
weight: male: ? (181-272 kg) n = ?; female: ? (91-1)
Range:
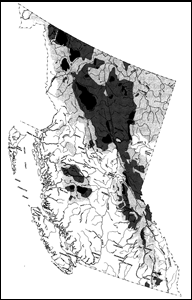
In British Columbia, Woodland Caribou inhabit boreal forests and mountain regions. The Northern ecotype is spread over a large area extending north and west of Prince George that can be roughly divided into two sections. The smaller part runs from around Kleena Kleene, northwest through the Hazelton Mountains and Tweedsmuir Park as far as the Nass River to the west of Mount Weber, and also extends over parts of the western Nechako Plateau. The larger section extends across much of northern British Columbia: its northern boundary is the Yukon border; the western edge is the southern St Elias Mountains and the east slopes of the Coast Mountains to about 56° 50’ latitude, through the Skeena Mountains onto the northeastern part of the Nechako Plateau east and northeast of Babine Lake; and the eastern boundary is the east slopes of the Rocky Mountains. The Boreal ecotype occurs at low densities from the eastern flanks of the Rockies as far as the Alberta border, except for a small area around Fort St John where there are no Caribou.
The range of the Mountain ecotype extends from its northern limit just south of Prince George to the international border, although recent work suggests that the northern boundary may be north of Prince George at Mount Mortifee in the Misininchinka Ranges. In the Rocky Mountains, the range extends from north of Mount Robson, south to the central Rockies west of Mount Columbia. These Caribou are also found on the east side of the Fraser River through the Quesnel Highlands south of Prince George, through the Monashee Mountains to Whatshan Lake, and also through the Columbia and Purcell mountains south to about Kitchener. In the Selkirk Mountains, Mountain Caribou occur as far south as Kaslo and the east side of northern Lower Arrow Lake, then there is a break in their distribution until it begins again in the southern Purcell Mountains, where it mixes with populations from northwestern Washington and northeastern Idaho.
A readily accessible viewing area for Woodland Caribou is at the top of the Salmo-Creston Pass, along Highway 3. They are often seen there in spring seeking salt on the road, and also in summer travelling through the area. Back-country visitors may also observe Caribou in Well’s Gray Provincial Park, Spatsizi Plateau Wilderness Provincial Park and Mount Revelstoke National Park.
Subspecies: Dawson's Caribou (Rangifer tarandus dawsoni) --
Extinct in British Columbia
Subspecies Description
Dawson’s Caribou is now extinct. There are only five museum specimens in the world, and none of the skulls are complete. All available materials, including skins and mounts, are in poor condition. From these specimens, researchers have come up with an uncertain description. It was a small Caribou, standing about one metre at the shoulder. Its body appears to have been mouse-grey, darker on top of the head and face, with a lighter neck and belly, a short mane, and probably without the clear brown-and-white markings found in other subspecies. The antlers were poorly developed, lacking the typical Caribou palmation, and it is uncertain if females possessed them.
Measurements
total length: male: 1,910 mm n = 1; female: 1,550 mm n = 1
tail vertebrae: male: 110 mm n = 1
hind foot: male: 385 mm n = 1;
shoulder: male: 1,050 mm n = 1
skull length: male: 350 mm (est.) n = 1
Range
Dawson’s Caribou is known with certainty to have lived only on the plateau around Virago Sound on the northern end of Graham Island in the Queen Charlotte archipelago, although an unverified skull was reported to have been found near a glacier at the mouth of the Skeena River on the mainland. Even on Graham Island, it was rarely seen, and the last three specimens were collected from a group of four in 1908. Dawson’s Caribou most probably became extinct either shortly after 1910 or in the early 1920s, although there were reports of tracks as late as 1935. The ancestors of these Caribou were most likely trapped on the Queen Charlotte Islands at the end of the last ice age when sea levels rose and cut off access to the mainland.