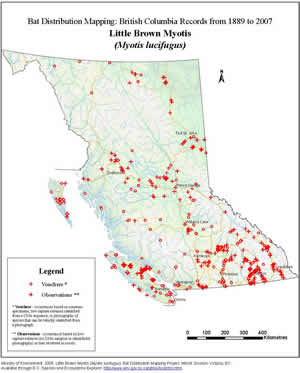
Mating occurs in late summer and early autumn before entering hibernation. Males do not breed in their first autumn. Although yearling females can breed in their first year, in British Columbia most delay reproduction until their second autumn. After a gestation period of 50 to 60 days, a single young is born; twins are extremely rare. Birth dates vary, determined in part by the date when females arouse from hibernation. In the southern Okanagan Valley, females give birth between the second week of June and the second week of July. Populations living at higher elevations and latitudes probably have their young later. There are no data on birth dates for coastal areas, but the earliest pregnancy record is 4 June from eastern Vancouver Island. Newborn young weigh 1.0-1.5 grams. They grow rapidly and by three weeks are capable of flying and eating solid food. Banding studies have revealed that this bat has a long life span with some individuals living more than 30 years.
|
The Little Brown Myotis emerges at dusk to feed. It is an opportunistic hunter that eats a great variety of insect prey. Robert Herd's research in the southern Okanagan Valley is the only study of food habits done in the province. He found that the Little Brown Myotis hunts in the valley and nearby hills in Ponderosa Pine forests, openings of trees, and over bluffs, lakes, rivers and irrigation flumes. Aquatic insects such as midges, caddisflies and mayflies are the major prey, although beetles, moths and other kinds of flies are also taken. The diet changes seasonally in response to insect abundance, with midges predominant in spring and caddisflies and mayflies most important in summer. The Little Brown Myotis is able to adjust its hunting techniques quickly to take advantage of insect concentrations. Most prey is captured in the air and eaten while flying. After an initial feeding period of 15 to 20 minutes, individuals occupy temporary night roosts near the day retreat. Night roosts are used most often when temperatures are cool (below 15°C) a protected night roost may help the Little Brown Myotis remain active so that it can digest its meal faster.
|
Summer roosts are in buildings and other man-made structures, tree cavities, rock crevices, caves and under the bark of trees. In summer the sexes live separately. Females congregate in nursery colonies that may contain hundreds or even thousands of individuals. Nursery colonies are in sites with hot (30-55°C), stable temperatures. Nursing females seem to prefer attics, but they will roost in almost any natural site that offers the environmental conditions that will promote the rapid growth of young. For example, a nursery colony of several hundred Little Brown Myotis was discovered in a small cave on the Grayling River in northern British Columbia. A hot (30°C), humid environment is maintained inside this cave by a natural hot spring.
Males rarely occupy nursery colonies. In summer they can be found roosting alone or in small colonies usually in sites that are cooler than the nurseries.
The Little Brown Myotis hibernates in caves and abandoned mines; it does not appear to hibernate in buildings. Some hibernacula in eastern Canada contain thousands of individuals. Hibernation records in British Columbia are limited to several old mines in the interior, each containing a few individuals. The whereabouts of most of the British Columbian populations in winter is unknown. Banding studies indicate that this bat will migrate 50 to 200 kilometres between hibernacula and summer roosts and if undisturbed it occupies the same sites year after year.
Before entering hibernation there is a period in late summer and early autumn when adults and young of the year make nocturnal flights through caves and mines, sites that are potential hibernacula. This swarming behaviour begins with the breakdown of the nursery colonies in late July and lasts until the bats enter hibernation. The role of swarming is not clear. It may be to familiarize young bats with the locations of potential hibernacula. Hibernation begins in September or October depending on the local climate. The Little Brown Myotis selects areas in the hibernaculum where there is high humidity (70-95%) and the temperature is above freezing (1-5°C). It may hibernate alone or in clusters. A change in temperature will waken this bat from hibernation to search for a new location in the hibernaculum. There are a few records of individuals moving to a new hibernaculum in mid winter. While hibernating, the Little Brown Myotis steadily burns up its fat reserves and loses about 25% of its weight by spring. In the interior of the province, hibernation probably lasts until April or early May, but in coastal regions this bat may arouse in late winter - it has been found feeding in coastal Oregon in mid March.
|
|